Global Atezolizumab Market, By Type (60mg/ml), By Application, and By Region - Trends and Forecast Analysis, 2021-2035
Publish Date: 2026-04-29 | Format: PDF | Category: Healthcare and Pharmaceutical | Pages: 354
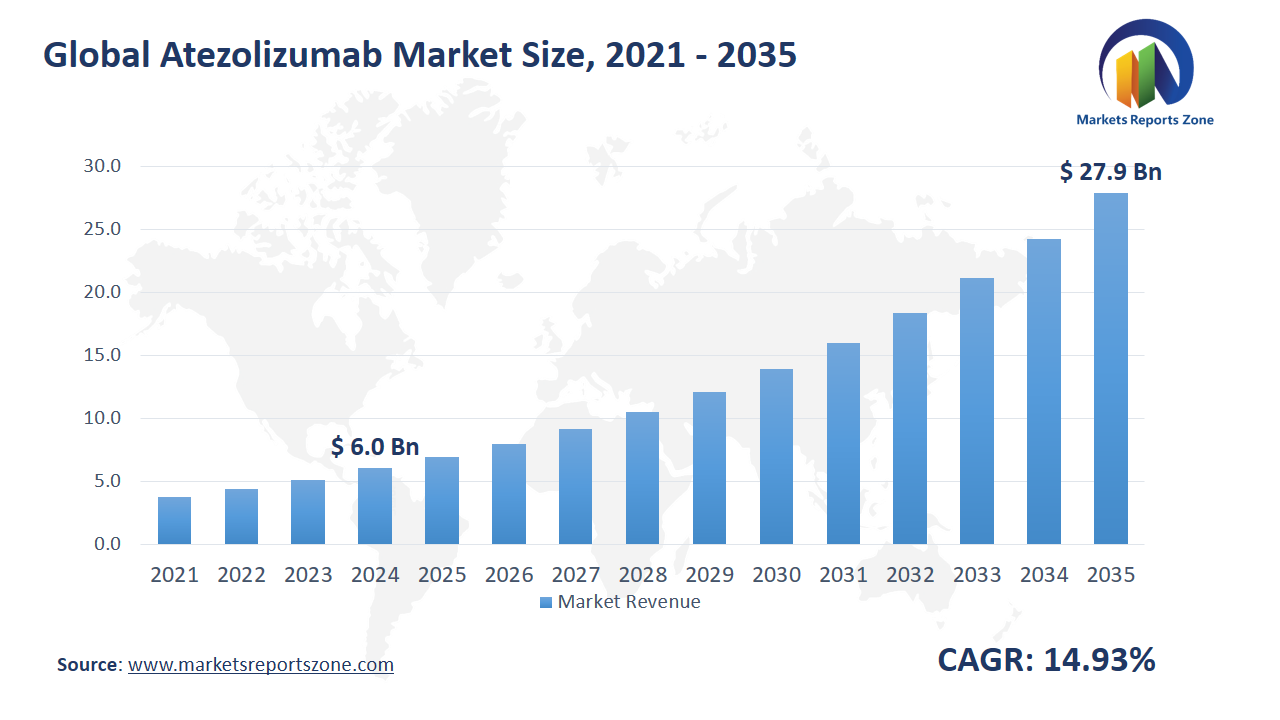
Global Atezolizumab Market Size is expected to reach USD 27.89 Billion by 2035 from USD 6.03 Billion in 2024, with a CAGR of around 14.93% between 2024 and 2035. The growth of the global Atezolizumab market is being driven by the rising cancer burden and increasing approvals of immunotherapy drugs. A steady rise in cancer cases has been reported worldwide. As a result, more patients are being treated with immune checkpoint inhibitors like Atezolizumab. Multiple drug approvals have also been granted in major markets, boosting availability. However, high treatment costs have been faced as a major restraint, limiting access in low-income countries. For instance, in some parts of Africa and Asia, Atezolizumab has been avoided due to affordability issues. On the other hand, new research trials have been conducted to explore Atezolizumab in early-stage cancers. Positive outcomes are being observed in neoadjuvant and adjuvant settings, creating room for early treatment adoption. Another opportunity is being seen through combination therapies. For example, Atezolizumab has been used with chemotherapy for advanced lung cancer and has shown improved survival. This approach is being widely explored in other solid tumors. Overall, more lives are being impacted as immunotherapies are integrated into cancer care. Though affordability challenges remain, new uses and combinations are being explored actively. In this evolving landscape, Atezolizumab is being positioned as a key component of next-generation oncology treatment strategies.
Driver: Cancer Surge Boosts Drug Demand
A major driver for the Atezolizumab market is the rising global cancer burden. As cancer cases have been steadily increasing across the world, the demand for advanced treatments has grown rapidly. More people are being diagnosed with cancers such as lung, bladder, breast, and liver at both early and advanced stages. In real life, hospitals in urban India have reported a sharp increase in younger patients with metastatic lung cancer, many of whom are now being considered for immunotherapy as a frontline option. Similarly, in Brazil, oncologists are seeing more patients with triple-negative breast cancer opting for immunotherapy combinations, as survival outcomes have improved. This trend is being observed in both developed and developing countries, where cancer screening programs have been expanded and awareness has increased. The result is more timely diagnoses and a larger pool of patients eligible for immune checkpoint inhibitors like Atezolizumab. As traditional treatments like chemotherapy and radiation alone often fall short in advanced cases, immunotherapy is being recommended more often by physicians. Patients and families are also actively seeking better outcomes and fewer side effects, pushing healthcare providers to include such therapies. As cancer numbers climb, the demand for effective drugs like Atezolizumab continues to rise.
Key Insights:
- Atezolizumab (Tecentriq) is approved for use in over 10 cancer types across more than 70 countries as of 2024.
- Genentech’s subcutaneous atezolizumab, launched in 2024, reduces administration time from 30–60 minutes to just 7 minutes for over 50,000 patients annually.
- Roche invested over $500 million in atezolizumab clinical development and expansion projects in 2023.
- More than 120,000 patients worldwide have been treated with atezolizumab since its first approval in 2016.
- In China, the incremental cost per QALY gained with atezolizumab over chemotherapy was $73,601.43, with a local willingness-to-pay threshold of $37,654.
- Chinese government price negotiations in 2022 reduced atezolizumab’s annual treatment cost by over 40%, benefiting more than 20,000 patients.
- Atezolizumab is listed in the national reimbursement drug lists of over 15 countries, raising its adoption rate to 38% among eligible cancer patients.
- The penetration rate of atezolizumab in first-line non-small cell lung cancer therapy reached 27% in 2023 across 500+ major hospitals worldwide
Segment Analysis:
The Atezolizumab market, segmented by type and application, is showing steady development with rising demand for targeted immunotherapies. The 60mg/ml formulation has been widely adopted in clinical settings due to its standardized dosing and ease of administration. Hospitals and infusion centers have preferred this concentration for its flexibility in creating customized doses based on patient needs. In oncology centers across South Korea, for instance, the 60mg/ml formulation has been routinely stocked for various treatment plans, reducing preparation time and streamlining care. In terms of application, strong growth has been observed in urothelial carcinoma treatment. Atezolizumab has been used as a valuable option for patients with advanced bladder cancer who have not responded well to platinum-based chemotherapy. In real practice, clinics in Poland have successfully used it to extend survival in elderly patients unfit for surgery. In the non-small cell lung cancer (NSCLC) segment, adoption has increased due to its role in first-line and maintenance therapy. In rural U.S. cancer centers, Atezolizumab has been used in patients with high PD-L1 expression, reducing hospital visits and improving quality of life. Across both cancer types, improved patient tolerance and better outcomes are being seen, making Atezolizumab a preferred immunotherapy option in multiple settings.
Regional Analysis:
The global Atezolizumab market has shown varied growth across five major regions—North America, Europe, Asia-Pacific, Latin America, and the Middle East & Africa—driven by differences in healthcare access, cancer prevalence, and policy support. In North America, especially the U.S., widespread insurance coverage and clinical trial networks have allowed quick adoption of Atezolizumab across cancer types. In Canada, hospitals in Ontario have started using it for maintenance therapy in lung cancer patients, reducing relapse rates. In Europe, countries like Germany and the Netherlands have integrated Atezolizumab into national cancer guidelines, ensuring early access. In Asia-Pacific, growth is rising sharply. Japan has incorporated it in multiple cancer protocols, while cities like Bangkok are seeing more private hospitals adopt it due to rising medical tourism. In Latin America, Brazil and Argentina have made slow but steady progress, with Atezolizumab being introduced in public hospitals under special cancer treatment schemes. Meanwhile, in the Middle East & Africa, access remains limited but is improving. In the UAE, private oncology centers are offering Atezolizumab to high-risk patients, while parts of South Africa are testing its cost-effectiveness. Across all regions, real-world success stories and survival benefits are fueling its gradual yet firm integration into standard cancer care.
Competitive Scenario:
Genentech, a subsidiary of Roche, continues to advance its Atezolizumab (Tecentriq) portfolio across multiple cancer types. In September 2024, the FDA approved Tecentriq Hybreza, a subcutaneous formulation combining Atezolizumab with hyaluronidase, offering patients a more convenient administration option. This development aims to enhance patient comfort and reduce clinic time. In hepatocellular carcinoma (HCC), the Phase III IMbrave050 study demonstrated that Tecentriq combined with Avastin significantly reduced the risk of cancer recurrence post-surgery, marking a pivotal advancement in adjuvant therapy. However, challenges have arisen; in November 2022, Genentech voluntarily withdrew the U.S. indication for Tecentriq in previously untreated metastatic urothelial carcinoma due to unmet endpoints in the IMvigor130 trial. Similarly, the company withdrew the accelerated approval for PD-L1-positive metastatic triple-negative breast cancer following the IMpassion131 trial's failure to meet its primary endpoint. Despite these setbacks, Genentech remains committed to expanding Tecentriq's applications. The ongoing SKYSCRAPER-01 trial is evaluating the combination of Tecentriq with tiragolumab in PD-L1-high metastatic non-small cell lung cancer, with interim results showing a trend toward improved overall survival. Additionally, Genentech is collaborating with BioNTech on Autogene cevumeran, an individualized mRNA cancer vaccine, exploring its potential in combination with Tecentriq to prevent cancer recurrence post-surgery. These initiatives underscore Genentech's dedication to enhancing cancer immunotherapy options.
Atezolizumab Market Report Scope
| Report Attribute | Details |
|---|---|
| Market Size Value in 2024 | USD 6.03 Billion |
| Revenue Forecast in 2035 | USD 27.89 Billion |
| Growth Rate | CAGR of 14.93% from 2025 to 2035 |
| Historic Period | 2021 - 2024 |
| Forecasted Period | 2025 - 2035 |
| Report Coverage | Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
| Regions Covered | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
| Countries Covered | U.S.; Canada; Mexico, UK; Germany; France; Spain; Italy; Russia; China; Japan; India; South Korea; Australia; Southeast Asia; Brazil; Argentina; Saudi Arabia; UAE; South Africa |
| Key companies profiled | Genentech |
| Customization | Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope. |
The Global Atezolizumab Market report is segmented as follows:
By Type,
- 60mg/ml
By Application,
- Urothelial Carcinoma Treatment
- Non-small Cell Lung Cancer (NSCLC)
By Region,
- North America
- U.S.
- Canada
- Mexico
- Europe
- UK
- Germany
- France
- Spain
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Saudi Arabia
- UAE
- South Africa
- Rest of Middle East and Africa
Key Market Players,
Frequently Asked Questions
Research Objectives
- Proliferation and maturation of trade in the global Atezolizumab Market.
- The market share of the global Atezolizumab Market, supply and demand ratio, growth revenue, supply chain analysis, and business overview.
- Current and future market trends that are influencing the growth opportunities and growth rate of the global Atezolizumab Market.
- Feasibility study, new market insights, company profiles, investment return, market size of the global Atezolizumab Market.
Chapter 1 Atezolizumab Market Executive Summary
- 1.1 Atezolizumab Market Research Scope
- 1.2 Atezolizumab Market Estimates and Forecast (2021-2035)
- 1.2.1 Global Atezolizumab Market Value and Growth Rate (2021-2035)
- 1.2.2 Global Atezolizumab Market Price Trend (2021-2035)
- 1.3 Global Atezolizumab Market Value Comparison, by Type (2021-2035)
- 1.3.1 60mg/ml
- 1.4 Global Atezolizumab Market Value Comparison, by Application (2021-2035)
- 1.4.1 Urothelial Carcinoma Treatment
- 1.4.2 Non-small Cell Lung Cancer (NSCLC)
Chapter 2 Research Methodology
- 2.1 Introduction
- 2.2 Data Capture Sources
- 2.2.1 Primary Sources
- 2.2.2 Secondary Sources
- 2.3 Market Size Estimation
- 2.4 Market Forecast
- 2.5 Assumptions and Limitations
Chapter 3 Market Dynamics
- 3.1 Market Trends
- 3.2 Opportunities and Drivers
- 3.3 Challenges
- 3.4 Market Restraints
- 3.5 Porter's Five Forces Analysis
Chapter 4 Supply Chain Analysis and Marketing Channels
- 4.1 Atezolizumab Supply Chain Analysis
- 4.2 Marketing Channels
- 4.3 Atezolizumab Suppliers List
- 4.4 Atezolizumab Distributors List
- 4.5 Atezolizumab Customers
Chapter 5 COVID-19 & Russia?Ukraine War Impact Analysis
- 5.1 COVID-19 Impact Analysis on Atezolizumab Market
- 5.2 Russia-Ukraine War Impact Analysis on Atezolizumab Market
Chapter 6 Atezolizumab Market Estimate and Forecast by Region
- 6.1 Global Atezolizumab Market Value by Region: 2021 VS 2023 VS 2035
- 6.2 Global Atezolizumab Market Scenario by Region (2021-2023)
- 6.2.1 Global Atezolizumab Market Value Share by Region (2021-2023)
- 6.3 Global Atezolizumab Market Forecast by Region (2024-2035)
- 6.3.1 Global Atezolizumab Market Value Forecast by Region (2024-2035)
- 6.4 Geographic Market Analysis: Market Facts and Figures
- 6.4.1 North America Atezolizumab Market Estimates and Projections (2021-2035)
- 6.4.2 Europe Atezolizumab Market Estimates and Projections (2021-2035)
- 6.4.3 Asia Pacific Atezolizumab Market Estimates and Projections (2021-2035)
- 6.4.4 Latin America Atezolizumab Market Estimates and Projections (2021-2035)
- 6.4.5 Middle East & Africa Atezolizumab Market Estimates and Projections (2021-2035)
Chapter 7 Global Atezolizumab Competition Landscape by Players
- 7.1 Global Top Atezolizumab Players by Value (2021-2023)
- 7.2 Atezolizumab Headquarters and Sales Region by Company
- 7.3 Company Recent Developments, Mergers & Acquisitions, and Expansion Plans
Chapter 8 Global Atezolizumab Market, by Type
- 8.1 Global Atezolizumab Market Value, by Type (2021-2035)
- 8.1.1 60mg/ml
Chapter 9 Global Atezolizumab Market, by Application
- 9.1 Global Atezolizumab Market Value, by Application (2021-2035)
- 9.1.1 Urothelial Carcinoma Treatment
- 9.1.2 Non-small Cell Lung Cancer (NSCLC)
Chapter 10 North America Atezolizumab Market
- 10.1 Overview
- 10.2 North America Atezolizumab Market Value, by Country (2021-2035)
- 10.2.1 U.S.
- 10.2.2 Canada
- 10.2.3 Mexico
- 10.3 North America Atezolizumab Market Value, by Type (2021-2035)
- 10.3.1 60mg/ml
- 10.4 North America Atezolizumab Market Value, by Application (2021-2035)
- 10.4.1 Urothelial Carcinoma Treatment
- 10.4.2 Non-small Cell Lung Cancer (NSCLC)
Chapter 11 Europe Atezolizumab Market
- 11.1 Overview
- 11.2 Europe Atezolizumab Market Value, by Country (2021-2035)
- 11.2.1 UK
- 11.2.2 Germany
- 11.2.3 France
- 11.2.4 Spain
- 11.2.5 Italy
- 11.2.6 Russia
- 11.2.7 Rest of Europe
- 11.3 Europe Atezolizumab Market Value, by Type (2021-2035)
- 11.3.1 60mg/ml
- 11.4 Europe Atezolizumab Market Value, by Application (2021-2035)
- 11.4.1 Urothelial Carcinoma Treatment
- 11.4.2 Non-small Cell Lung Cancer (NSCLC)
Chapter 12 Asia Pacific Atezolizumab Market
- 12.1 Overview
- 12.2 Asia Pacific Atezolizumab Market Value, by Country (2021-2035)
- 12.2.1 China
- 12.2.2 Japan
- 12.2.3 India
- 12.2.4 South Korea
- 12.2.5 Australia
- 12.2.6 Southeast Asia
- 12.2.7 Rest of Asia Pacific
- 12.3 Asia Pacific Atezolizumab Market Value, by Type (2021-2035)
- 12.3.1 60mg/ml
- 12.4 Asia Pacific Atezolizumab Market Value, by Application (2021-2035)
- 12.4.1 Urothelial Carcinoma Treatment
- 12.4.2 Non-small Cell Lung Cancer (NSCLC)
Chapter 13 Latin America Atezolizumab Market
- 13.1 Overview
- 13.2 Latin America Atezolizumab Market Value, by Country (2021-2035)
- 13.2.1 Brazil
- 13.2.2 Argentina
- 13.2.3 Rest of Latin America
- 13.3 Latin America Atezolizumab Market Value, by Type (2021-2035)
- 13.3.1 60mg/ml
- 13.4 Latin America Atezolizumab Market Value, by Application (2021-2035)
- 13.4.1 Urothelial Carcinoma Treatment
- 13.4.2 Non-small Cell Lung Cancer (NSCLC)
Chapter 14 Middle East & Africa Atezolizumab Market
- 14.1 Overview
- 14.2 Middle East & Africa Atezolizumab Market Value, by Country (2021-2035)
- 14.2.1 Saudi Arabia
- 14.2.2 UAE
- 14.2.3 South Africa
- 14.2.4 Rest of Middle East & Africa
- 14.3 Middle East & Africa Atezolizumab Market Value, by Type (2021-2035)
- 14.3.1 60mg/ml
- 14.4 Middle East & Africa Atezolizumab Market Value, by Application (2021-2035)
- 14.4.1 Urothelial Carcinoma Treatment
- 14.4.2 Non-small Cell Lung Cancer (NSCLC)
Chapter 15 Company Profiles and Market Share Analysis: (Business Overview, Market Share Analysis, Products/Services Offered, Recent Developments)
- 15.1 Genentech
Report ID:
231
Published Date:
April 2026
Trusted by more than 10,500 organizations globally



Infaluble Methodology
Customization
Analyst Support
Targeted Market View